Most people think of a bone as a rigid, even dead structure like a skeleton in a museum. To Sarah Dallas, Ph.D., it’s a fascinating matrix of active cells at work. And she has the images to prove it.
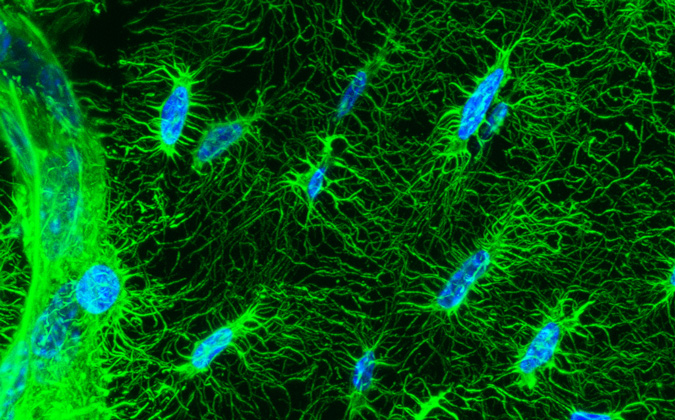
With the use of high-tech microscopes at the UMKC Confocal Imaging Core, Dallas has pioneered the method of taking time-lapse images of live bone cells over a period of days and assembling them into movies that provide novel insights into how the living cells work to form the mineralized bone matrix.
Dallas serves as director of the imaging core and the muscle and bone phenotyping core, which supports the research efforts of a diverse range of investigators at UMKC. Her research specializes in techniques for live-cell imaging and is now moving toward the imaging of cells within live animals.
Two pieces of the core’s equipment, a confocal microscope and multiphoton microscope, are used to capture fixed cell images and images of live animal cells. These microscopes, obtained through funding from a series of National Institutes of Health grants, are the highly-specialized machines that Dallas used to develop her unique approach to the imaging of cell functions and their dynamic actions in assembling bone tissue.
“The study we’re doing is basic science research,” Dallas says. “It’s a step back from clinical applications, but we’re trying to identify potential approaches for preventing bone loss.”
Dallas is an internationally recognized expert in her work with a particular cell called an osteocyte. Her imaging projects have proven the cell, once considered to be completely inactive because it is embedded within the bone matrix, to be extremely dynamic. Recently, osteocytes have been found to control the activities of two other important bone cells: osteoblasts, which form bone, and osteoclasts, which resorb bone.
Bones are in a constant state of turnover, where bone is being resorbed and replaced. With age or disease, that delicate process becomes imbalanced and results in bone loss, known as osteoporosis.
In addition to developing a better understanding of the fundamental underpinnings of bone loss, Dallas’ work could also have implications for treating bone disorders such as osteogenesis imperfecta, or brittle bone disease, a genetically inherited disease that affects children and makes their bones even more susceptible to fractures.
“In terms of clinical applications, the more we know about osteocytes, the more we’ll be able to tap into their potential for developing drugs for preventing bone loss in osteoporosis and other bone conditions,” Dallas says.
Dallas’ research projects have three primary targets: looking at bone cell dynamics, exploring how the extracellular bone matrix is assembled and studying the role of osteocytes in age-related bone loss.
Her work has also expanded to looking at the interaction of bone and muscle in aging. As bone mass declines throughout the aging process, so does muscle function. In collaboration with muscle biologists, she is exploring the crosstalk between bone and muscle with the idea that the tissues may be linked in some way so that they actually share signals that lead them to deteriorate together.
Dallas discovered her fascination with research and bone biology while working on her undergraduate degree at the University of Birmingham, U.K., and her Ph.D. at the Royal Veterinary College in London. While she was doing post-doctoral work on bone-related cancers at the University of Texas Health Science Center in San Antonio, a particular project led her to explore the role of protein-regulating growth factors on the bone matrix.
She returned to the U.K. to work at the Welcome Trust Center for Cell Matrix Research in Manchester, where Dallas says she thought she would stay and finish her research career. That changed when her postdoctoral supervisor from San Antonio, Lynda Bonewald, Ph.D., one of the world’s leading bone scientists, called in 2011 and recruited Dallas to join her mineralized tissue research group at UMKC.
Dallas’ research projects, while continually funded by the NIH since 2001, have become internationally known. Last year, a crew from the Japanese public television network, NHK, came to UMKC to interview Dallas and film a segment for an eight-part documentary on the human body that featured experts from throughout the world.
“Some of our work directly relates to osteoporosis and some to inherited connective tissue disorders like osteogenesis imperfecta,” Dallas says. “The rest is basic science that feeds into all of these. Understanding the fundamental mechanisms of how these cells work feeds into the future for figuring out potential treatments for diseases.”



